Authors
Meg Hooton, President, IQVIA Biotech
Todd Matthews, MBA, Senior Director, Corporate Alliances, IQVIA
Dean Giovanniello, RPH, MBA, Sr. Principal, Financial Institutions & Brand and Commercial Strategy Consulting Services, IQVIA
In our previous post, we explored how a well-crafted Target Product Profile (TPP) lays the foundation for a compelling value story that resonates with investors. But a strong vision alone isn’t enough. To truly capture investor confidence, early-stage biotech companies must also demonstrate how they plan to bring that vision to life efficiently, strategically and with a clear understanding of risk. That’s where a detailed plan for collecting evidence, including a Clinical Development Plan (CDP), becomes indispensable.
Why Development Strategy Is Central to Biotech Funding
Investors are increasingly discerning. They want to see more than promising science; they want a roadmap that shows how a company will navigate regulatory hurdles, reach proof-of-concept (PoC), and ultimately deliver commercial value. The CDP is the operational backbone of that roadmap.
A robust CDP enables teams to model and compare multiple development pathways, each with distinct timelines, costs and risk profiles. For early-stage companies facing capital constraints and scientific uncertainty, this flexibility is essential.
“The CDP should be treated as a living document, one that evolves with emerging clinical data, regulatory feedback and shifts in the competitive landscape.”
Start Early, Stay Ahead
One of the most overlooked advantages in biotech development is timing. Initiating the CDP during lead candidate selection, not after, is a strategic advantage. Early planning ensures that trial design, indication selection and regulatory strategy are aligned from the outset, reducing the risk of costly course corrections later.
This early start also allows companies to integrate the CDP with the TPP, ensuring that clinical development is directly tied to commercial goals and stakeholder expectations.
Choosing the right path
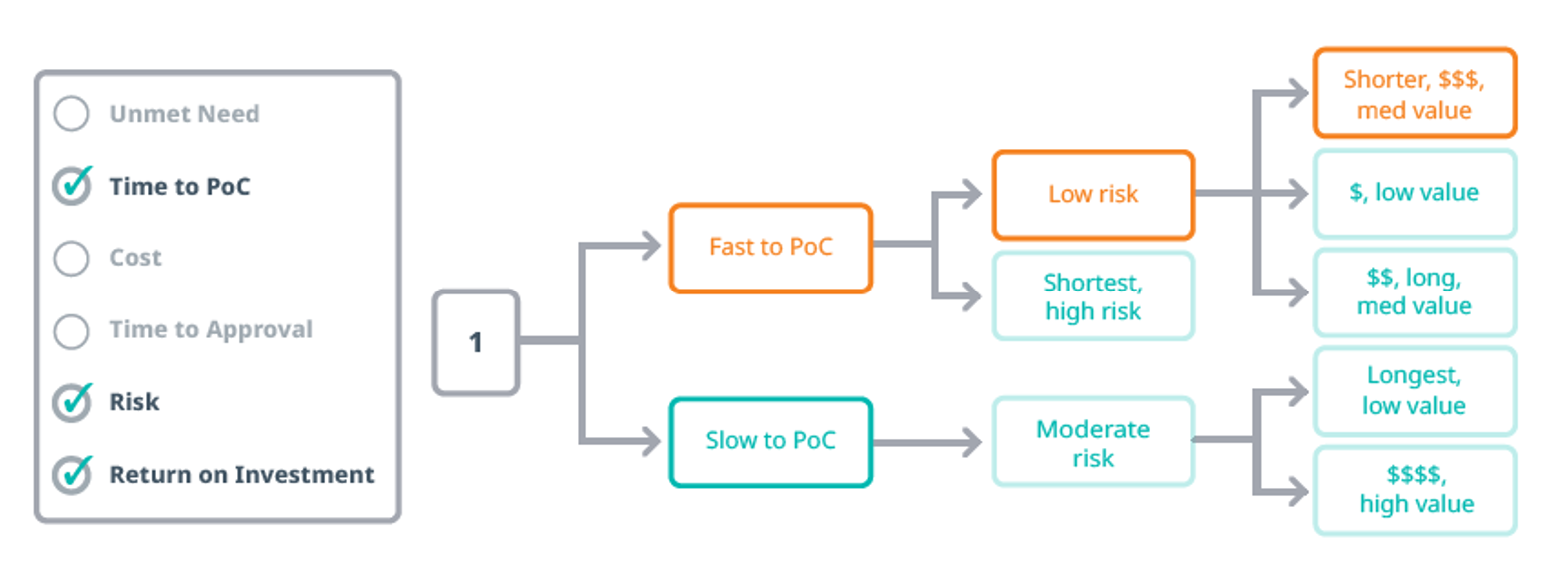
Mapping the Path: Time, Cost, and Risk
Every development path involves trade-offs. To make informed decisions, biotech leaders must evaluate three core variables:
- Time: How long will it take to reach a critical value inflection point, such as PoC or regulatory approval?
- Cost: What resources are required to get there, and how do they scale across different trial designs?
- Risk: What is the probability of success at each phase, and how does it vary by indication, drug type and trial complexity?
By modeling these variables, companies can identify viable alternatives and prioritize development options that align with their strategic goals and investor expectations.
Strategic Indication Selection
Choosing the right indication is one of the most consequential decisions in early-stage development. Different indications carry varying levels of risk, cost and market potential. For example:
- Rare diseases often benefit from expedited regulatory pathways and smaller trials, but face recruitment challenges and limited market size.
- Cardiovascular programs have seen rising success rates, making them attractive for development.
- Inflammation/ immunology where there is significant competition, leading to demand for higher standards of efficacy and clearer differentiation.
- Infectious diseases, on the other hand, have experienced declining approval probabilities, requiring careful risk assessment.
By integrating real-world data and historical success rates, companies can select indications that maximize asset value and align with investor priorities.
Cross-Functional Collaboration Drives Success
A robust CDP is built through cross-functional collaboration. Engaging clinical, regulatory, commercial and operational teams early ensures that the development strategy is both scientifically rigorous and commercially grounded. This alignment helps avoid missteps and ensures that the plan reflects the realities of execution and market entry.
The CDP as a Living Document
The CDP should be treated as a living document, one that evolves with emerging clinical data, regulatory feedback and shifts in the competitive landscape. This adaptability strengthens investor confidence and supports agile decision-making throughout development.
Communicating Strategy with Confidence
A well-developed CDP is more than an internal planning tool, it’s a communication asset. It demonstrates to investors that the company has:
- A clear understanding of development trade-offs
- A data-driven approach to risk management
- A strategy aligned with regulatory and commercial milestones
By integrating adaptive trial designs, go/no-go criteria, and real-world evidence, the CDP becomes a dynamic tool that supports both conservative and accelerated strategies.
Final Thoughts
Choosing the right development path is one of the most powerful ways early-stage biotech companies can de-risk their venture and attract capital. Through strategic scenario modeling, indication selection and competitive analysis, biotech leaders can build a Clinical Development Plan that not only guides internal decisions but also inspires investor confidence.
In our next post, we’ll explore how to quantify and communicate the value of your development strategy using data-driven insights, specifically, how to leverage eNPV to strengthen your case with investors.
Visit us at https://www.iqviabiotech.com/visionaries to learn more.
References
Clinical Development Success Rates and Contributing Factors 2011–2020. Biotechnology Innovation Organization (BIO), Informa Pharma Intelligence, and QLS Advisors, Biomedtracker and Pharmapremia, 2021, https://go.bio.org/rs/490-EHZ-999/images/ClinicalDevelopmentSuccessRates2011_2020.pdf
Aitken M, Roland A, Kleinrock M. Global Trends in R&D 2025: Progress in Recapturing Momentum in Biopharma Innovation. March 2025. IQVIA Institute for Human Data Science. https://www.iqvia.com/insights/the-iqvia-institute/reports-and-publications/reports/global-trends-in-r-and-d-2025

 Search
Search