Addressing COVID-19 impact. Ensuring progress.



A new reality for clinical trials
In a few short months, COVID-19 has transformed how healthcare works. It has diverted expertise and resources and changed the way patients are diagnosed and treated. It has disrupted the development and delivery of medicines.
The result is a new environment for clinical trials. And while some issues may resolve in the short term, others may signal a more permanent shift.
That is why IQVIA Biotech deployed a task force dedicated to ensuring patient safety and trial continuity. We are here to help customers navigate today’s challenges and ensure your business, and your commitment to patients, remains strong.
Standardized risk assessments. Custom solutions.
IQVIA Biotech’s Clinical Development COVID-19 task force quickly mobilized to develop a standardized approach to risk assessment for both in-flight and new trials.
Customers receive custom reports detailing risks and mitigation strategies to support trial continuity and patient safety, and to help you continue to drive healthcare forward - today.
Strategic regulatory support
The global pandemic has changed the regulatory landscape and guidance continues to evolve. Clinical trial sponsors should consider COVID-19 mitigation measures and robust risk benefit assessment to avoid the need for costly changes later on.
In-flight studies may require protocol and study modifications to ensure patient visits and dosing continue. IQVIA Biotech’s regulatory experts offer guidance to support this flexibility in trial delivery and the safety of enrolled patients.
Whether you need submission strategy, amendments for home visits, support for EC and IRB submissions, or other global regulatory documents, our global regulatory experts can guide you.


Supported patients. Trial continuity.
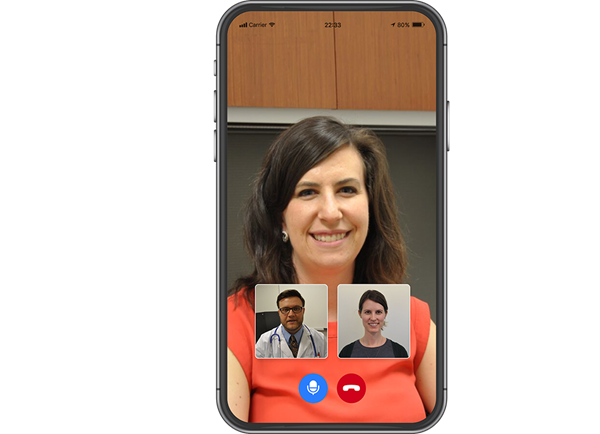
Telemedicine empowered.
Travel restrictions and limitations on face-to-face interactions may necessitate moving to a fully remote or hybrid decentralized trial model to help ensure continuity and compliance. IQVIA Study Hub is a cloud platform that allows patients and study teams to securely connect in a televisit. It also gives patients the ability to upload medical history or patient reported outcomes easily from their tablet, smartphone or web browser.
Patients need to stay home. Meet them there.
Some trial patients may require home-based care. Through our network of partners, IQVIA offers at-home support by trained nurses and phlebotomists, helping to reduce the risk of infection in healthcare settings.
We further support in-home visits by shipping lab supplies and investigational product to the patient or the home health nursing partner.
Clinical trial resources
IQVIA Biotech is proud to be part of the global effort against COVID-19 by accelerating development of treatments and vaccines. We are currently supporting multiple customers running COVID-19 studies.
Our team remains committed to deliver flexible clinical trial solutions, and seamlessly integrates IQVIA capabilities and resources to keep trials moving forward in this new reality.



Centralized monitoring for safety and continuity
Whether through televisits, site visits or home-based care, IQVIA Biotech is effectively monitoring sites and patients through remote monitoring. By leveraging centralized monitoring, we are pivoting clinical trials from traditional monitoring to proven risk-based monitoring solutions, allowing a rapid mitigation strategy for COVID-19 impact. This approach includes
- Study risk assessments to identify the optimum remote monitoring strategy to support patient safety and data quality
- Our global scale to rapidly deploy site performance indicators and remote mitigation steps
1,000 virtual meetings
Investigator Meetings conducted by IQVIA since 2018

Maintain timelines
As more interactions are moving to virtual settings, it is critical to ensure timelines and productivity are maintained.
IQVIA’s experienced facilitators are a seamless extension of your study team, providing content creation and delivery as well as logistics and technical management.
Supported by professional trainers, behavioral strategists, medical writers and technical producers, our team has facilitated more than 1,000 virtual meetings in the past two years earning a >90% satisfaction rating.

