Persuading investors of your product's value involves a balancing act between the importance of cost, time, risk and commercial return.
As an emerging biopharma, you have selected your candidate and articulated your product vision through the creation of a target product profile – so you know where you are going. You have planned your development pathway, balancing time, cost, and risk at specific decision points – so you know how you are getting there. Both steps are vital in capturing the attention of investors, and have been outlined in the first two blogs in the series.
This post (3rd of 3) takes you to the next level by helping you to optimize and demonstrate the value of your approach. Creating an early and accurate forecast model; understanding time, cost, risk, and revenue in more detail; and valuing your program in terms of risk-adjusted or expected net present value (eNPV) will all be critically important when convincing key stakeholders to join you.
Creating a single, unifying metric: Expected net present value
There are several key variables that you need to be aware of in the process of optimizing your drug development approach:
- Cost: Development and commercial costs
- Time: Critical path duration of the plan
- Risk: Probability of technical success
- Commercial return: Potential market share and revenue
Expected net present value (eNPV) is a robust methodology that provides a convincing way to weigh different approaches. Using eNPV as a unifying metric means that you can compare different molecules, indications, or design decisions and see the impact of trade-offs. For example, you might evaluate the impact of changing potential market share and revenue by looking at a different indication.
Emerging biopharma are under pressure to show value
While demonstrating value is important for any organization developing new drugs, the pressure is more acute for emerging biopharma companies. Emerging biopharma companies are smaller, have less cash in hand, and have a greater need for convincing investors at any given point in the drug development pathway.
Data-informed planning for robust decision-making
With access to the right data and domain knowledge, it is possible to understand the impact of various factors on crucial outputs.
|
Domain knowledge |
Trial design |
Development plan |
Key drivers of risk |
Market share analogues |
|
Data |
Development experience |
Clinical trials data |
Industry benchmarks |
Sales databases |
|
Outputs |
Costs |
Time |
Risk |
Commercial return |
While all the critical success factors are important, modeling the interplay between these factors can be challenging. For example, indications for rare diseases generally have a higher probability of success than higher-prevalence diseases but also a smaller pool of potential patients which may slow enrollment. Plans to offset with a higher price point can present a major conundrum for investors and ultimately for payers. New indications for proven drugs and biologics are more likely to succeed than new molecular entities, but may face much stiffer competition in the marketplace. Additionally, patient-centricity also plays a role although it may not be imperative for investors at an early stage.
Scenario generation and modelling can support choosing the best clinical design from among many options, allowing you to see what happens to eNPV, for example under conservative, standard and aggressive approaches to time and cost trade-offs.
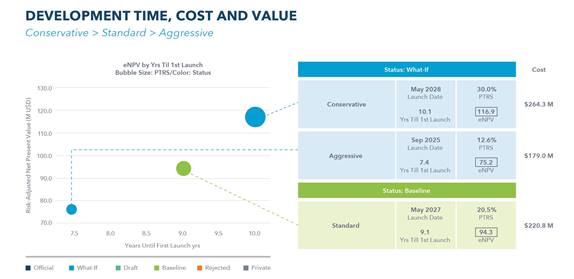
The three blogs in the Capturing the attention of investors series have each taken a different part of the whole. The first, on ‘articulating your product vision’, looked at the importance of understanding business drivers and ensuring their alignment with product goals. The second, on ‘choosing the right development path’, explored design decisions, alternatives and trade-offs. And this, the third, ‘the value of your approach’, focused on optimizing cost, time, risk and/or commercial return. These three together should put you, as an emerging biopharma company, on the road to success.
To hear experts talk in more depth and listen to them answer audience questions, go to Capturing the attention of investors for the recorded webinar, supported by a slide presentation.
More about the Author:
- Bruce Basson
Senior Scientific Advisor
Bruce has worked as a biostatistician in the pharmaceutical industry for over 20 years across numerous therapeutic areas. One of his areas of expertise is leading development teams through scientific evidence generation plan strategy and optimization workshops. He is also a specialist in technical due diligence and asset value estimation using net present value.
Contributor:
- Tara Rheault, Ph.D., MPH
Senior Therapeutic Strategy Director, Drug Development Lead
Tara Rheault has over 16 years of cross-functional leadership experience across drug discovery and development. Dr. Rheault started her career as a medicinal chemist at GlaxoSmithKline where she was the inventor of the now marketed drug Tafinlar. Dr. Rheault then moved on to global project leadership for early development projects, including portfolio and governance management. She then transitioned into late stage clinical development as a clinical program and study leader. Dr. Rheault has continued to build her expertise at IQVIA in the development of integrated commercial and R&D strategies, helping clients articulate their differentiated value story along with a robust plan to achieve their product goals.

